Surfactants are amphiphilic molecules possessing a hydrophilic (water-loving) and a hydrophobic (water-avoiding) region. As a result, surfactants are surface-active-agents that can adsorb onto fluid/fluid interfaces and reduce the surface tension, facilitating wetting, foaming, and/or emulsification of pharmaceuticals, food products, detergents, personal care products, cosmetics, and oilfield chemicals.

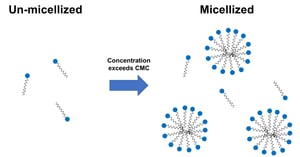
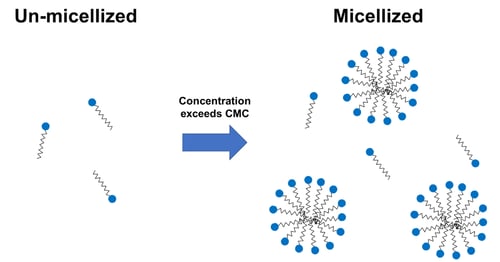
The critical micelle concentration (CMC) is the concentration of surfactants above which micelles form and all additional surfactants added to the system will form micelles. It is a fundamental and important parameter in surfactant science, as well as in applications involving detergents, cosmetics, personal care products, oilfield chemicals, pharmaceuticals, and food products.
When a surfactant’s concentration in solution exceeds the critical micelle concentration (CMC), it self-assembles into spherical micelles, helping with solubilization of oils and dirt, as well as enhancing the stability of emulsions and foams. The CMC is affected by conditions like temperature, salinity, pH, and solvent composition.
Properties of a surfactant solution that exhibit an abrupt change at the CMC include the viscosity, surface tension, conductivity, light scattering, turbidity, and osmotic pressure. The CMC can be determined by measuring the property and locating the break in a plot of its value vs. concentration of surfactant (see image). Viscosity measurements can be used to calculate the CMC.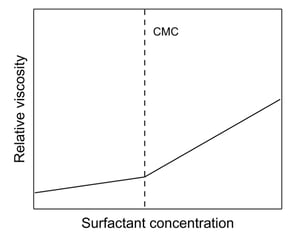
A challenge in determining the CMC is the low sensitivity for the measured property, like viscosity. To obtain more accurate values, measurements are typically conducted for multiple concentrations below and above the CMC. The VROC® viscometers, with their sensitive, high throughput, and small volume capabilities, can be used to easily calculate the CMC of surfactant solutions under different conditions, and can be used to complement other methods like tensiometry.
In our application note (Determination of the critical micelle concentration of a surfactant from viscosity measurements), we utilized the VROC® initium one plus for viscosity measurements to identify the CMC of sodium dodecyl sulfate (sodium lauryl sulfate), an anionic surfactant with heavy use in household & personal cleaning products and in gel electrophoresis techniques. In addition, we calculate the CMC under different solvent compositions (aqueous or aqueous-glycerol solvent) to show the robustness of our VROC® instrument.
Download the application note for the full data analysis, or contact us to speak with one of our rheology experts about how to use viscosity measurements to calculate CMC.
Written by: Chrystian Ochoa, PhD, RheoSense Application Scientist