Monoclonal antibody therapy is a form of immunotherapy that uses monoclonal antibodies (mAb) to bind monospecifically (targeting the same antigen) to certain cells or proteins, which if successful will stimulate the patient's immune system to attack those cells. mAb therapeutics are often used for targeted treatment of cancer, autoimmune diseases, numerous infectious diseases and even Alzheimer’s disease. Since 1985, approximately 100 monoclonal antibodies have been designated as drugs (Manis et. al).
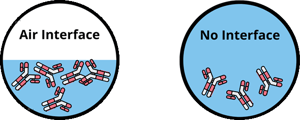
Air interface is the phenomena when it comes to air becoming mixed with measurements causing readings to be impacted. Proteins are amphiphilic molecules that in aqueous solutions tend to move to the air-liquid interface to minimize potential energy. The accumulated protein molecules at the interface form a viscoelastic film, which affects the apparent viscosity of the solution. (Baek, GEN, 2015) In the past, the effect of air interface has been shown for bovine serum albumin (BSA) and lysozyme solutions, however the air interface is also problematic when measuring viscosity of mAb solutions.
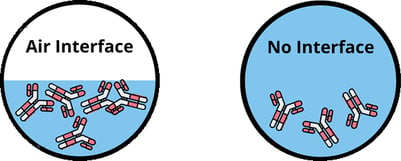
These air-liquid interfaces can lead to aggregation of your mAb, which can cause instability of your formulation and even effect the drug product quality. Surface adsorption and aggregation at these air interfaces can occur during manufacturing, purification, formulation, storage, and clinical administration. To minimize mAb aggregation, many formulators add surfactants to their protein formulations, however few quantitative characterizations of the surface mechanical properties or molecular adsorption at the relevant interface have been performed to guide the selection and composition of surfactant. (Langmuir 2020, 36, 7814−7823)
When it comes to viscosity measurements, if air is impacted, often you will see a greater percent in error, higher viscosity results, and overall inaccuracies in your viscosity measurements. The interface between air and protein solutions can lead to major errors in viscosity measurements of protein solutions, which can mislead your formulations and result in possible disqualification of otherwise successful candidates. The protein molecules are adsorbed at the air interface to minimize overall free energy and rearrange to form a viscoelastic film, which in turn affects the apparent viscosity of the solution. The film leads to erroneously high viscosity measurements of protein solutions by an order of magnitude.
Conventional rheometers measure inaccurate and higher viscosities of protein solutions because of the presence of the air interface. For example, viscosity measured with a cone and plate rheometer erroneously produces high viscosity measurements of samples subjected to low shear rates, because it assesses the interfacial layer of protein and not the protein solution itself. (Baek, GEN, 2015)
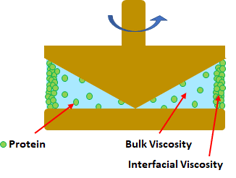
RheoSense provides viscometer solutions free of the air interface problem, which is ideal for viscosity measurement of mAb solutions. Our viscometers are powered by a patented VROC® (Viscometer-Rheometer-on-a-Chip) technology. VROC technology combines microfluidic and MEMS (Micro-Electro-Mechanical Systems) technologies to measure small sample volume dynamic viscosity of mAb solutions over a wide dynamic range of operation free of air-interface problem. VROC® viscometers are enclosed instruments, meaning there is no fluid-air interface impacting your measurements. All VROC® chips (formally listed in USP chapter <914> as rectangular slit) are fully enclosed in their own chip housing to prevent any air interface.
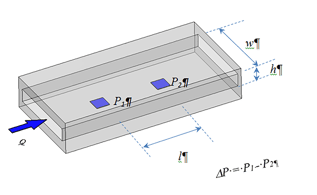
Compared to conventional viscometers and rheometers, our viscometers offer several exceptional advantages:
- Small sample size requirement (26µL – 100µL)
- Cost-saving
- Characterization of both Newtonian & non-Newtonian fluids
- Enable high shear rates simulating injection
- Prevent evaporation and contamination of samples
- Render high throughput due to a simple flow-through design
Learn more about our VROC powered viscometers and their additional benefits here.
Written by: Eden Reid, RheoSense Senior Marketing Associate