Syringeability and injectability are both important parameters in the field of biopharmaceuticals, especially when dealing with the complex formulations of biologics and concentrated protein solutions. While they may appear to be interchangeable, they are in reality two very different parameters. Syringeability is the ease of withdraw of a solution from vial to syringe, whereas injectability is "a term related to the ease of parenteral administration of a dosing solution, and includes dose preparation, dose administration, ergonomics related to these procedures, pain of injection, and other adverse events at the injection site". (Watt et. Al, 2018)
Syringeability includes such factors as ease of withdrawal, clogging and foaming tendencies, and accuracy of dose measurements. Injectability includes pressure or force required for injection, evenness of flow, and freedom from clogging (i.e., no blockage of the syringe needle). (Cilurzo et. Al, 2011)
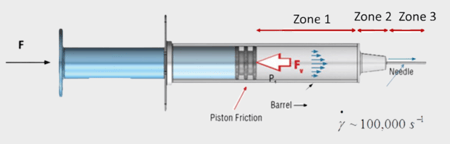
The viscosity of a solution has a direct impact on both the syringeability and the injection force or injectability. Needle geometry also directly impacts both parameters. the difference between the two is most evident with high concentration formulations where viscosity would be higher and therefore play a bigger role (especially with higher gauge needles). The force required to uptake higher viscosity solutions might be very high, so choosing the right the plunger and barrel combo for your syringe might effect the syringeability. Whereas with injectability the pressure drop across the syringe needle dominates, meaning it is essentially how much force is needed to push the sample out at a given injection rate.
In protein therapeutics, major resources are dedicated toward the development of injection formulations for:
- Sustainable release
- Subcutaneous injection
Both the force to withdraw a solution from the vial and the injection force required for an injectable solution are important in early formulation and development of an auto-inject system. The viscosity of a protein solution has a direct impact on the injection force or injectability of the solution. Understanding the viscosity at the shear rate inside your needle is crucial to estimating an accurate injection force, which has a direct impact on both efficacy and patient experience. It is as important to measure the viscosities of the candidate drugs and drug injectability at a very early stage of development for qualification in order to maximize the return on investment and mitigate the risk of developing a drug that is impossible to deliver.
VROC® technology allows viscosity measurements up to 200,000 s-1 and eliminates the guesswork out of injectability studies in early stage formulation development by allowing direct and accurate estimation of the injection force.
Download our Predicting Injection Force eBook for a complete guide to predicting injection force from theoretical foundation to protocols you can follow to estimate the injection force of Newtonian and non-Newtonian fluids.
Written by: Eden Reid, RheoSense Senior Marketing Associate