Cell culture media is essential for many in labs in academia, national laboratories, and the biotechnology industry. Cell culture media is vital for studying cellular processes, developing new drugs, and creating regenerative medicine (Yao and Asayama, 2017). Even emerging fields such as lab grown meat products require new types of media be developed and characterized (Gaydhane et al, 2018).
Cell culture media is a complex fluid filled with nutrients required for cells to grow, proliferate, and differentiate. Media is specifically designed to maintain the ideal nutrients, hormones, pH, and osmotic environment necessary for cell growth. In particular, the amount of glucose and other nutrients can modify the rheology of the media itself, which can impact cell growth and differentiation. We know different types of cells prefer different types of media. For example, many mammalians cell lines are grown and maintained in Dulbecco’s Modified Eagle Media (DMEM) or Ham’s or F12 media, while insect cells are grown in Grace’s medium. Characterizing the differences between formulations is vital for developing new products and for maintaining quality control for existing product lines.

When discussing cell culture media, we sometimes hear, “viscosity is not relevant to my work”, however we know this is not the case as cell culture formulation can be quite relevant to viscosity. We know that viscosity can be used to characterize differences between cell and tissue culture media. When formulating cell culture media it is important to keep in mind that out of the lab setting cells are never in a static environment, so it is important to provide a formulation that is more realistic and beneficial to cell growth. Shear force on cells helps them to grow most realistically.
Through a series of scientific test, RheoSense, investigated the viscosity of Fetal Bovine Serum (FBS) Dulbecco′s Modified - Eagle′s Medium (DMEM), and F12 cell culture media formulations. Our investigation of the viscosity of a few DMEM showed that subtle changes in viscosity can be detected based on the different amounts and types of nutrients used in the formulations. Our investigation of F12 media formulations showed that filtering the media with a 0.2 µm filter does not impact media viscosity, there is not a difference in viscosity between purchased and homemade DMEM/F12 media, and the viscosity of DMEM/F12 is equivalent to the DMEM component in the formulation.
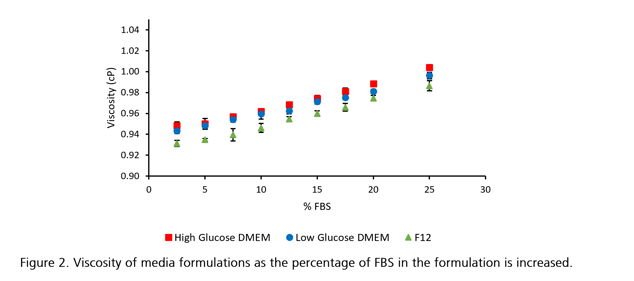
Cell culture media are sometimes supplemented with serum to provide cells with even more nutrients such as hormones, growth factors, and fat-soluble vitamins depending on cell type and application. A commonly used serum used in cell and tissue culture is fetal bovine serum (FBS). We investigated the viscosity of FBS as well as FBS supplemented DMEM and F12 media formulations and were able to show that filtering FBS does not change its viscosity and that FBS supplementation does change the viscosity of cell and tissue culture media formulations. We also observed that the incorporation of D-glucose in cell culture media formulations can influence their rheological behavior.
The data collected in our investigations demonstrates that viscosity can be used to further characterize differences between cell and tissue culture media both for research and development purposes as well as for quality control and manufacturing. Characterizing formulation-to-formulation differences and batch-to-batch differences will enhance product development when creating new media formulations which become vital for multiple applications in the future, including cell and tissue culture, lab grown meat products, and regenerative medicine.
Download our ebook to learn more about cell culture media formulation and the importance of viscosity in cell culture media! 
Written by: Zachary Imam, PhD Biomedical Engineering