A key consideration during the drug development process is delivering the final product to the patient. Whether that happens orally, topically or via injection into the patient, a drug must be delivered to be effective. The injectable delivery pathway is common for antibody-based therapeutics (Allmendiger et al 2014). These formulations involve high concentration protein formulations greater than 150 mg/mL in some cases. Protein formulations are thermodynamically metastable, which means the measurable characteristics of these solutions change over time. At these high concentrations, a minor change in concentration can lead to a dramatic change in viscosity.
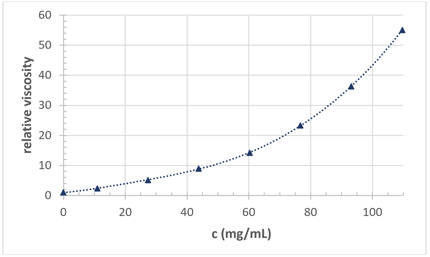
The rheological behavior of proteins in aqueous solutions change as they age. These changes are driven by intermolecular interactions between the proteins in solution. Proteins in solution do not exist in a static environment. They diffuse under Brownian motion even at refrigerated temperatures leading to collisions between protein molecules. These collisions can result in a change in microstructure or spatial arrangement which can be observed as changes in viscosity.
The viscosity of a protein solution has a direct impact on the injection force or injectability of the solution. Protein solutions can be prepared many months before they are used, and the age of a protein formulation can dramatically impact its application and functionality. For injectability, metastability means that the force required to deliver the protein formulation will change as the formulation ages. Specifically, for injectable drugs, the ageing process can increase the force required for injection, which is a critical metric for the viability of a drug candidate. A high injection force can be painful for a patient and can make delivery impossible.
Different formulation conditions including ionic strength or excipient choice can impact the storage stability of protein formulations. The inclusion of excipients can greatly impact the ageing process of proteins. Excipients such as arginine-HCl can reduce the viscosity of protein formulations (Dear et al 2019). Commonly, protein formulations increase in viscosity as they age, which can directly influence their useability and deliverability. A formulation that meets the cutoff viscosity when it is first formulated may not be injectable once it has been stored for a few months. Understanding the influence of formulation ageing on injectability is vital during the drug development process for this reason. A longer formulation shelf life means that the solution is viable long after it has been formulated, shipped, and delivered to the patient.
In a recent application note, we demonstrated how the incorporation of arginine-HCl into protein formulations reduced initial viscosity, inhibited flocculation, and kept viscosity stable for longer. In our newest application note, we investigate the viscosity changes of two model protein formulations as they age and use the viscosity and shear rate data to estimate the injection force of these formulations. Additionally, we demonstrate how the incorporation of arginine-HCl into formulations not only increases storage stability, and therefore solution shelf life, but it also reduced injection force.
Download the full application note to learn more about how monitoring viscosity as injectable protein solutions age is vital for understanding how they will behave in the time between formulation and delivery.
Written by: Dr. Zachary Imam, RheoSense Research Scientist